FDA Checker
Create FDA-compliant PDFs for regulatory submissions with ease. Automated checking, bookmarking, and table of contents generation.
Last updated March 2026

Browse our free online tools. Mapsoft's PDF Hub offers 60+ free PDF tools in your browser. The FDA Checker plug-in below provides regulated submission compliance checking inside Acrobat — the eCTD-specific validation is not available in the online tools.
Streamline FDA Compliance for PDF Submissions
Preparing PDF documents for FDA regulatory submissions requires strict adherence to formatting guidelines. Mapsoft's FDA compliance tools work together with Adobe Acrobat to ensure your documents meet all FDA requirements -- from proper bookmarking and table of contents structure to page formatting and document organization.
Our step-by-step approach takes the guesswork out of FDA compliance, letting you focus on your submission content while our tools handle the technical formatting requirements.
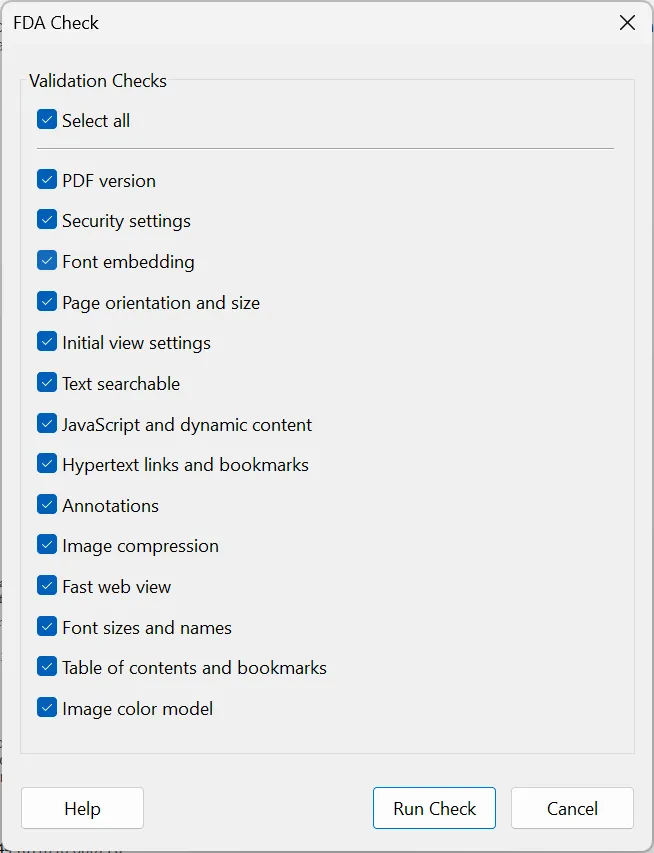
FDA Compliance Tools
A suite of Acrobat plugins designed for regulatory document preparation.
Bookmarker
Automatically create and manage PDF bookmarks from document structure. Generate compliant bookmark hierarchies that meet FDA submission standards.
TOCBuilder
Generate FDA-compliant tables of contents for your submission documents. Automatically builds hyperlinked TOC pages with proper formatting and structure.
Compliance Checking
Automated validation of PDF documents against FDA submission guidelines. Identify and resolve compliance issues before submission.
Document Structure
Ensure proper page numbering, section organization, and document hierarchy that complies with FDA electronic submission requirements.
Page Formatting
Control page sizes, margins, and layout to match FDA specifications. Ensure consistent formatting across all sections of your submission.
Submission-Ready Output
Produce final PDF documents that are fully ready for FDA electronic submission, with all compliance checks passed and proper formatting applied.
FDA Submission Guidelines Covered
Our tools address the key technical requirements for FDA PDF submissions.
Bookmark Requirements
FDA submissions require properly structured bookmarks that mirror the document's table of contents. Our Bookmarker tool automates this process entirely.
Table of Contents
Generate hyperlinked tables of contents that meet FDA formatting standards, with proper indentation, numbering, and page references.
Document Navigation
Ensure reviewers can easily navigate your submission with properly linked sections, cross-references, and consistent document structure.
File Size & Format
Optimize PDF files for submission, ensuring they meet size requirements and use acceptable fonts, images, and compression settings.
Resources
User Guide
Complete documentation covering FDA Checker features, compliance checks, and configuration options.
Download
Download Bookmarker and TOCBuilder to start preparing your FDA-compliant submissions.
Purchase
Ready to buy? Visit the Mapsoft store to purchase licenses for our FDA compliance tools.
System Requirements
Adobe Acrobat
Adobe Acrobat Pro or Standard (DC, 2020, or 2017). Not compatible with Adobe Acrobat Reader.
Operating System
Windows 10 or Windows 11 (64-bit recommended). Both 32-bit and 64-bit Acrobat installations are supported.
Licence
Perpetual licence with free maintenance period. See licence details and upgrade policy.
See It in Action
Related Articles
Creating FDA-Compliant PDF Submissions
A step-by-step guide to creating FDA-compliant PDFs using Adobe Acrobat, Mapsoft Bookmarker, and Mapsoft TOCBuilder. Covers text searchability, font embedding, bookmarks, and TOC creation.
PDF/A: The Archival PDF Standard
An in-depth guide to PDF/A — the ISO 19005 standard for long-term document archiving. Covers conformance levels, requirements, prohibited features, and conversion workflows.
“The products deliver high quality for a nominal price, backed by the best technical support I’ve encountered.”
Michael Fero — University of Washington
Need Something More?
If this product doesn't quite meet your requirements, we can help. With over 30 years of Adobe development expertise, our team can modify existing products or build entirely custom solutions tailored to your specific workflow. Whether you need additional features, integration with your systems, or a completely bespoke tool, get in touch to discuss your needs.
We also welcome suggestions for new features and new product ideas. If we hear enough requests for a particular capability, it moves up our development priority list — so your feedback directly shapes what we build next.
Beyond off-the-shelf plug-ins, we also build enterprise PDF solutions on the Adobe PDF Library — server-side document processing, generation, validation, conversion and redaction pipelines for high-volume environments. If your need is bigger than a desktop tool, our PDF Library team can build it.
Need Help with FDA Submissions?
$129
One-time purchase · includes 1 year of updates · perpetual licence
